|
How Caffeine is Extracted
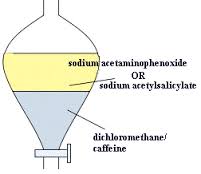
Caffeine can be extracted using many common solvents such as Benzene, chloroform, trichloroethylene, and dichloromethane. The three main processes that have proven more successful than these common solvents are water, supercritical Carbon Dioxide and organic solvents. |
|
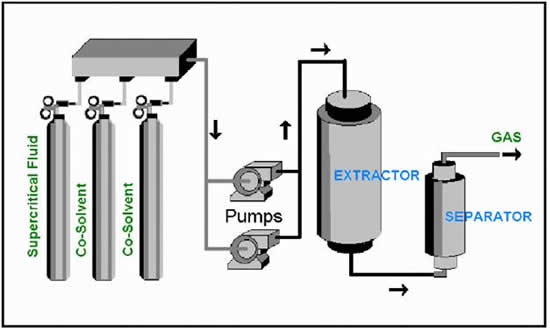
Supercritical Carbon Dioxide Extraction:This extraction uses carbon dioxide because it is safer than other organic solvents. To begin, carbon dioxide is forced through green coffee beans at a temperature above 31.1 °C and a pressure above 73 the standard atmospheric pressure. Under these conditions, carbon dioxide enters a “supercritical” state, which has gas-like properties, that allows the carbon dioxide to enter deep into the beans. As it enters deep into the beans it maintains its liquid like properties, allowing it to dissolve 97-99% of the caffeine in the beans. The carbon dioxide that has dissolved the caffeine is sprayed with high-pressure water to remove the caffeine. This water is than filtered out, allowing the caffeine to be isolated into its purest form, usually a white powder.
|