|
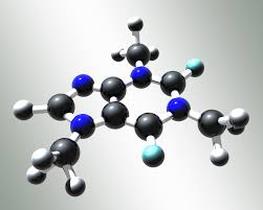
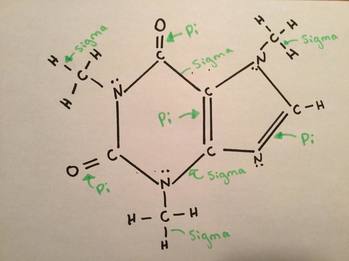
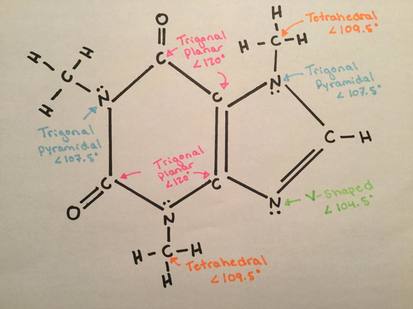
Sigma and Pi BondsSigma and pi bonds are covalent bonds which are end to end overlapping of orbitals. “A sigma bond is created by an end to end overlap of atomic orbitals” (Nelson Chemistry 12). “A pi bond is created by the parallel overlap of atomic orbitals, usually having p orbitals” (Nelson Chemistry 12). This picture shows where sigma and pi bonds are found in the caffeine molecule.
|
|